![[Image]](images/pict2.jpg)
Elimination reactions produce unsaturated compounds. Alcohols undergo elimination to produce an alkene. Alkyl halides undergo eliminations as well. Using a secondary alcohol we will perform an acid-catalyzed elimination which is second order (E1), meaning that a carbocation is the intermediate and the rate of reaction is dependent on how quickly the carbocation can be formed. As soon as the carbocation is formed, the rest of the elimination reaction occurs very quickly. Starting with 2-pentanol the major product should be the formation of 2-pentene, although a description of how other alkenes could be formed is included. Since neither GC or IR allow us to distinguish these other alkenes, we presume that the major (perhaps only) product we isolate will be the 2-pentene, although it is possible that some of the other alkenes may be present. An IR will be performed showing that the alcohol group has been removed.
Dehydration of an alcohol can follow either the E2 or the E1 reaction mechanism. However, in each case, acid is required as a catalyst. Because the OH- (hydroxide) ion is a poor leaving group (it is a strong base), we perform the reaction in acid to produce water (HOH) as a leaving group, since it is a much weaker base. The reaction is initiated by adding a strong acid, such as H2SO4 (not HCl or HBr because the resultant anion (Cl- or Br-) would react and produce the alkyl halide) to the mixture. Acid will protonate the hydroxyl group of the alcohol to produce an alkyloxonium ion; R-OH2+) to allow for a water molecule to be the leaving group. Once the alkyloxonium ion is produced, the reaction mechanism that is followed depends on the nature of R-group. For a primary alcohol, such as 1-pentanol, the dissociation of water, if it occurred, would produce the very unstable and hard to form 1° (primary) carbocation. As a result, we would project that elimination via an E1 mechanism for a primary alcohol will be difficult to occur. As a result, reaction would be expected to proceed via the E2 elimination mechanism for any primary alcohol, and end up producing an ether instead of an elimination product. However, for a secondary alcohol, such as 2-pentanol, dissociation of water produces the more stable 2° (secondary) carbocation. Because water is not a strong base, the competing E2 mechanism would be slower, which allows for the E1 mechanism to proceed faster for 2-pentanol.
Dehydration of an alcohol can follow either the E2 or the E1 reaction mechanism. However, in each case, acid is required as a catalyst, because OH- is a strong base, it is a poor leaving group:
![[Image]](images/pict2.jpg)
Adding a strong acid, such as H2SO4, to the mixture allows protonation of the -OH group to give water as a leaving group. Once this protonation occurs, the mechanism that is followed depends on the nature of the R group. As mentioned above, 1-pentanol (a 1° alcohol), dissociation of water would produce the very unstable 1° carbocation, so we would project that elimination via an the E1 mechanism (with carbocation intermediate) will not occur. As a result, reaction would be expected to proceed via the E2 elimination mechanism. However, for 2-pentanol, dissociation of water produces the more stable 2° carbocation. Because water is not a very strong base, the competing E2 mechanism will be slow, which will allow the E1 mechanism to proceed faster for 2-pentanol. The mechanism below depicts reaction by E2 mechanism to product, in a single, concerted step, an elimination, producing an alkene. The only product, via an E2 reaction mechanism, would be 1-pentene.
![[Image]](images/pict1.jpg)
In 2-pentanol, dissociation of water will give a more stable 2° carbocation. Because water is not a strong base (it is not readily attracted to one of the H atoms on the b-carbon), the E2 elimination mechanism will be slow, which will allow the E1 mechanism to be faster for the 2-pentanol.
![[Image]](images/pict0.jpg)
Note that all three possible products are shown. The actual product mixture will be determined by gas chromatography to determine which of the three possible products is produced. You will determine the percent composition of this mixture.
There is one more event that is possible in a reaction that involves carbocation intermediates, and is even a possibility with the E2 reaction shown for 1-pentanol, and that is rearrangement. The 2° carbocation produced in the E1 reaction of 2-pentanol may rearrange to give the more stable 3° carbocation as well. If this occurs, two more possible products can be envisioned:
![[Image]](images/pict3.jpg)
It is also possible that the 1° alkyloxonium ion formed by the protonation of 1-pentanol has the potential to rearrange via a 1,2-hydride shift (which kicks off the water molecule as the leaving group). This reaction will give the same secondary carbocation initially produced with 2-pentanol as the starting material.
![[Image]](images/pict4.jpg)
The only way to know for sure whether or not a rearrangement has taken place is to determine the identities of the products, and the relative amounts of each product, in each reaction mixture must be determined. We will use gas chromatography to do this, very much like we did in Experiment IV.
Safety: 2-pentanol is a volatile and flammable liquid as well as being an irritant. No flames will be allowed in the lab. Wear gloves while handling these chemicals. Concentrated sulfuric acid is strongly corrosive and toxic -- wear gloves while handling it, and be sure to wash your gloves and your hands immediately after handling it. Sodium sulfate is an irritant -- gloves are recommended. The alkene products are all highly flammable, and have irritating vapors -- avoid breathing their vapors.
Set up the reaction in the following order.
Allow the solution to cool to room temperature before proceeding further.
Set up a simple distillation (no thermometer is needed so you must seal the still head using a polyethylene seal and cap). The 100-mL flask containing the reaction mixture is used as the distillation pot.
Attach a pre-massed 50-mL round bottom flask as the receiver. The receiver flask is placed in an ice water bath to keep it cold during the reaction and distillation because the resultant alkene has a low boiling point. It is good practice to weigh the receiving flask with a glass stopper in it, since you will need to have the stopper in it when you determine the mass of your collected product. Begin heating the mixture (you can use a high setting for the heating mantel at first to get the reaction started). Once the mixture begins to boil (drops start to collect in receiver vessel), adjust the heat setting to give a slow, but steady, boil in the distillation flask. Continue the distillation for about an hour, or until no more distillate is collected, whichever comes first.
After the distillation, dispose of the strongly acidic waste contents of the distillation pot in the appropriate liquid waste container, being very careful not to spill any of it, but disposing of the boiling stones in the solid waste container.
Determine the mass of the collected crude product (remember, that your collected material contains both water and organic material).
Wash the isolated liquid in a separatory funnel using some of the 5% NaOH(aq). Collect only the organic layer from the separatory funnel. (Which layer in the separatory funnel is aqueous, and which is organic? How can you tell?)
Add the organic phase from your Separatory Funnel extraction to a clean and dry 50- or 100-mL round-bottom flask. Usually you will use a 19/22 ground-glass flask, since the normal 14/20 flasks are kept in the kits and used by other labs. In order to dry the organic layer you will use anhydrous sodium sulfate (you should use about 2.0 g of anhydrous Na2SO4 to dry your chemical; that amount of anhydrous salt will absorb about 2.5 g of water). Unless you can clearly see a water layer in your flask, the 2.0 g above should be sufficient to dehydrate your organic liquid.
Store your properly labeled flask (with organic phase and dehydrating salt) until the next lab period. Attach a ground-glass stopper to a round-bottom flask for storage. You cannot just use parafilm as your distillate will dissolve the parafilm.
Obtain your flask containing the distillate mixture from the previous lab period (this liquid material should contain your desired alkene product as well as some 2-pentanol which also distilled over). When you perform a distillation to re-purify your desired chemical, be certain that no salt crystals from the bottom of the drying flask is added to the distillation pot (containing a couple boiling stones). Since the salt crystals contain absorbed water, heating them would release the water, and your drying procedure would be for naught.
When you re-distill your dried (anhydrous; absence of water) organic material, be sure you use a thermometer so that you can monitor the boiling point of the distillate. Collect the distillate as before (for this distillation, use the 4-mL collection vial from your organic chemistry kits). Place the collection vial in an ice bath as before and collect your anticipated product, monitoring the temperature, until the 4-mL vial is full (or until the temperature rises above 41oC) but slightly below the ground-glass opening.
Do the following analyses of your product to help characterize it. Remember, that any one physical property (e.g., boiling point) cannot be used to identify a chemical. However, when several physical properties or analyses are performed, you can be more certain that the chemical being analyzed is the chemical you are anticipating when more of the values agree with that chemical.
Alkene (colorless) + Br2 (orange) --> dibromoalkane (colorless)
For any reactions, show the complete reaction of reactants and products in your notebook and formal report (if written).
As we have seen in lecture, and in Part A of Experiment VI, dehydration of an alcohol can follow either the E2 or E1 reaction mechanism, with protonation of the alcohol as the first step of each reaction. (Which reaction proceeds faster?) With E1 (like SN1 reactions), however, the carbocation intermediate can undergo rearrangement to give a more stable carbocation, which then undergoes the final deprotonation step to give a double bond:
![[Image]](images/image7.gif)
The carbocation rearrangement may involve either an alkyl shift (a methyl group is the alkyl group which shifts in the example depicted above, but more complex alkyl groups may also shift) or a hydride (H:–) shift. The requirements for a shift are: (1) that the carbocation be able the achieve a conformation which allows overlap between an empty p orbital on the cation carbon, and the s orbital (on a hydrogen atom) or the small lobe of the sp3 orbital (on a carbon atom of an alkyl group) of the shifting group; and (2) that the new carbocation (e.g, 3o>2o>1o) be more stable than the original.
In the dehydration of pinacol, there are two possible mechanisms, one which involves a potential carbocation rearrangement, and one without:
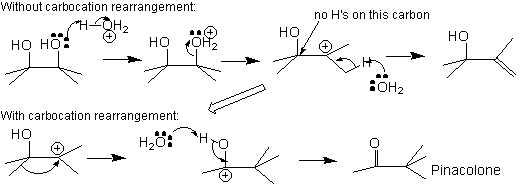
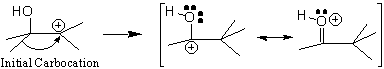
Which one is more likely? Remember that a rearrangement will occur only if it gives a more stable carbocation. Look carefully at the carbocation formed after a potential rearrangement. Moreover, if a resonance structure(s) can be drawn, potential rearrangements will be much more likely.
Resonance stabilizes a cation better than inductive effects alone can, so the new cation, as a result of resonance contributors, is more stable than the initial carbocation, and pinacolone is the major product of this reaction.
Safety: Pinacol is a flammable solid and an irritant -- no flames will be allowed. Wear gloves while handling it. Concentrated sulfuric acid is strongly corrosive and toxic -- wear gloves while handling it. Be sure to wash your gloves and your hands immediately after handling any of the above chemicals. Anhydrous magnesium sulfate is an irritant -- gloves are recommended. The Pinacolone is a flammable liquid, and have irritating vapors -- avoid breathing their vapors.
Put 6.0 g of pinacol and 30 mL of 3 M H2SO4 in a 100-mL round bottom flask. Set up a simple distillation, with the 100-mL flask containing the reaction mixture serving as the distillation pot. Use a labeled 50-mL round bottom flask as the receiver. Once the mixture begins to distill, adjust the heat to keep a moderate boil. The distillate will separate into two layers, and aqueous layer and your product (you need to decide which layer is your product, based on density). Stop the distillation when the water-insoluble layer is no longer increasing in volume (this should take about 20 minutes, or so, from the time the first drop of distillate is collected).
Dispose of the strongly acidic waste material remaining in the distillation pot into the appropriate waste container, being very careful not to spill any of it. As in Part A, you should consider neutralizing the distillate using NaHCO3. After neutralization, wash the crude product (from your receiver flask) with 15 mL of deionized water using a separatory funnel. You need to determine which of the two phases in the separatory funnel is your desired product, and how you can best recover this material. Dry your organic product using anhydrous magnesium sulfate (adding the appropriate amount to dry the organic material, as described in Part A).
Redistill the dry, crude product, using a pre-massed receiver flask (e.g., a conical vial) and monitor for boiling point. Determine the mass, volume and refractive index of the re-distilled product. Compare the refractive index to the literature value. Show your sample to the instructor. Discard of the material in the appropriate waste container.
| Compound | MW | Amount | mmol | mp | bp | Density | ηD | msds |
|---|---|---|---|---|---|---|---|---|
| 2-pentanol | 88.15 | 10.0 mL | --- | -75 | 119.3 | 0.8092 | msds | |
| 2-pentene (and other alkenes) | 70.13 | --- | --- | -140 | 37 | 0.65 | --- | msds |
| Br2 (in CH2Cl2) | 159.808 | --- | -7.25 | 58.78 | 3.11 | msds | ||
| Compound | g/mol | grams or mL | 10-3 M | oC | oC | g/mL | ηD | msds |
none