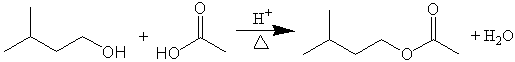
You will perform an acid-catalyzed esterification reaction to make a common ester present in many different fruits. This ester, isopentyl acetate is one of the chemicals in bananas. Using standard laboratory procedures, you will isolate and characterize your product. Initially a reflux will allow the reaction to proceed for about an hour. The product of reaction will be collected following a simple distillation, washed and dried prior to isolating it in more pure form in the next lab. Your product will be collected and analyzed using IR, refractive index, density, etc.
Esterification is a straightforward reaction that utilizes several key techniques in synthetic organic chemistry. One direct approach, known as the Fischer esterification reaction, involves the acid-catalyzed condensation of an alcohol and a carboxylic acid, yielding an ester and water. Esters can also be formed by the reaction of the alcohol with the acid chloride rather than the acid itself. Or, the acid anhydride may be used instead of the acid. In this experiment, we will create the ester isopentyl acetate (banana oil) via the Fischer esterification reaction.
Esters are an important group of carboxylic acid derivatives. Many esters occur naturally. They often compose a significant fraction of the fragrant oils of fruits and flowers. Fats and oils are another type of natural ester; they are actually mixtures of glycerol triesters, made of long-chain fatty acids and glycerol (glycerin: 1,2,3-propanetriol). Waxes are esters of long-chain carboxylic acids and long-chain primary alcohols. Lactones are cyclic esters that arise when a carboxyl group and an alcohol group in the same molecule condense to form an ester. Synthetic esters are also important and found in many places, including the polyesters known as Dacron, prepared from terephthalic acid and ethylene glycol, and Kodel.
Emil Fischer (1852-1919) was a German scientist who received his Ph.D. from the University of Strasbourg, studying with Adolf von Baeyer. He taught at Erlangen, Wurzburg, and Berlin Universities. Fischer's work essentially laid the foundation of modern biochemistry. Fischer was the first to synthesize phenylhydrazine, which was an important reagent in his work on elucidating the structures of most of the carbohydrates. During a three-year period beginning in 1891, Fischer established not only the basic structures, but also the configurations of all the known sugars. In the process, he developed a method to represent the three-dimensional molecular structures in two-dimensional drawings. These structures have become known as Fischer projection formulas. This work by Fischer led directly to proving the existence of the asymmetric carbon atom, a concept proposed by Van't Hoff and Lebel in 1874.
In addition to carbohydrate chemistry, Fischer did extensive work on the chemistry of purine and compounds having purine as their nucleus. Purine is one of the two nitrogen base ring systems present in DNA.
Fischer was also active in the area of protein chemistry. He demonstrated that amino acids are the basic subunits from which proteins are constructed. He also devised methods for the synthesis of many of the known amino acids. Perhaps his most ingenious contribution was the 'lock and key" hypothesis of how proteins bind with substrates of complementary shapes. This work ultimately led to our understanding of how enzymes, the catalysts of biochemical reactions, function.
Regarded as the greatest organic chemist of his time, Emil Fischer became the second chemist to receive the Nobel Prize (1902). Fischer committed suicide in 1919 following the death of his wife and the loss of two of his three sons.
The Fischer esterification is an equilibrium reaction whereas other esterification routes do not involve an equilibrium. To shift the equilibrium to favor the production of esters, it is customary to use an excess of one of the reactants, either the alcohol or the acid. In the present reaction, we will be using an excess of the acetic acid, because it is cheaper and easier to remove than the alcohol (note the similar boiling points of the alcohol and acetate). Another way to drive a reaction toward its products is to remove one of the products as it forms. In this experiment, we will remove the water formed in the reaction by adding silica beads directly to the reaction vessel and by using a drying tube with drying agent (calcium chhioride), which prevents the introduction of water.
The following figure shows the reaction for this experiment:
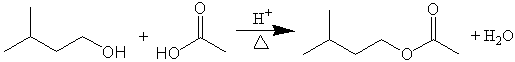
The mechanism for this reaction involves the nucleophilic addition of the alcohol to the carbonyl group of the protonated acid, followed by elimination of a proton. The tetrahedral intermediate is unstable under the acidic conditions of the reaction and undergoes dehydration to form the ester.
The key steps of this mechanism
involve the following:
Because esters can be hydrolyzed under acidic or basic conditions, it is not a good idea to stop this reaction before the acid catalyst has been neutralized. Hydrolysis is the breaking of the ester apart back to the acid and the alcohol. This reaction can be useful; if the hydrolysis is carried out under basic conditions, it is referred to as a saponification. Saponification is an irreversible reaction in which one mole of base is consumed per mole of ester to generate the carboxylic acid anion.
Assemble the following apparatus:
The apparatus will be heated using a heating manifold placed on top of a stirring hot plate (hot plate is not used to provide heat, only to stir the contents of the reaction flask). Secure the reaction setup to a ring stand using a three prong clamp attached to the condenser column, so that the reaction vessel (after the reaction is completed) can be easily elevated and cooled in the air.
The reaction components are added as follows.
After the 60-minute reaction is complete, turn off the heating mantel and elevate the reaction flask to allow it to cool to room temperature.
Carefully decant (pour) the liquid part of the reaction container using a funnel into a Separatory Funnel (make certain that the silica gel beads and stirring bar remain in the reaction container). Remove the stirring bar and discard the silica gel beads into the solid waste container.
After your reflux is finished, and the liquid reaction mixture has been added to the Separatory Funnel, you will need to do washes to neutralize the sulfuric and acetic acid in the mixture. To do these washes, use three 20-mL portions of 5% sodium bicarbonate which will neutralize the acid in the mixture. The separatory funnel is capped and then shaken vigorously, with frequent venting (carbon dioxide gas is produced). After each of the three washing steps, allow the layers separate and then remove and discard the lower aqueous layer. The upper layer will be your isopentyl acetate and the lower layer will be the aqueous bicarbonate and water layers are collected into another flask and eventually discarded in liquid waste.
The bicarbonate washings of the ester product are followed by a single 20-mL wash using distilled water to remove any bicarbonate ions that may still be mixed with product. After the water layer (bottom layer) is removed, the isopentyl acetate is transferred to an 100-mL round bottom flask (use 19/22 ground glass flask).
Your collected isopentyl acetate must be dried using a dehydrating agent. You should use anhydrous sodium sulfate (Na2SO4) to dry your chemical. Add about 2 grams of the anhydrous sodium sulftate (this amount of anhydrous sodium sulfate is enough to bind over 2.5 grams of water).
Store your liquid product, with drying agent, until the next lab period. Prior to distillation of your product during the second day, the anhydrous isopentyl acetate is carefully poured or decanted away from the solid sodium sulfate in the drying flask. Put your dried ester into a clean, and dry, 50- or 100-mL round bottom flask for distillation.
Purification of the crude isopentyl acetate is performed by distillation. The entire product is added to a 50-mL round bottom flask, containing several boiling stones to prevent super-heating and bumping of the crude isoamyl acetate during distillation. A simple distillation is performed to collect the isoamyl acetate. The flask is heated in using a heating manifold. The isopentyl acetate will boil and condense and will be collected. This distillation will significantly purify the material, but not if there is alcohol or water present. You should collect your material when you have a product at the appropriate boiling point of the ester.
The boiling point of the liquid should be measured as the isopentyl acetate distills. The thermometer is placed in the distillation head. A lot of heat is lost to the atmosphere when using this very small equipment, so a thermometer placed in the distillation head is a pretty good indicator of the boiling point.
An analysis of your product should include the following:
The spectra are the IR's for isopentyl alcohol (isoamyl alcohol) and isopentyl acetate (an ester).
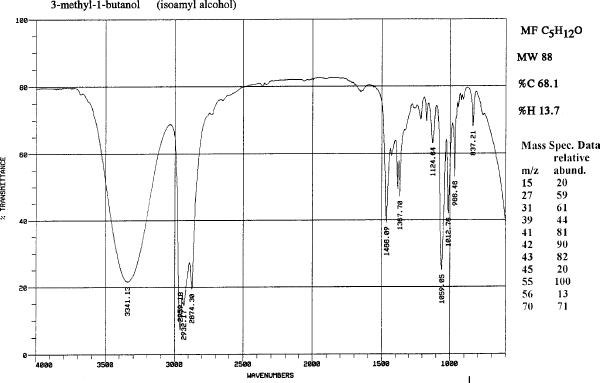
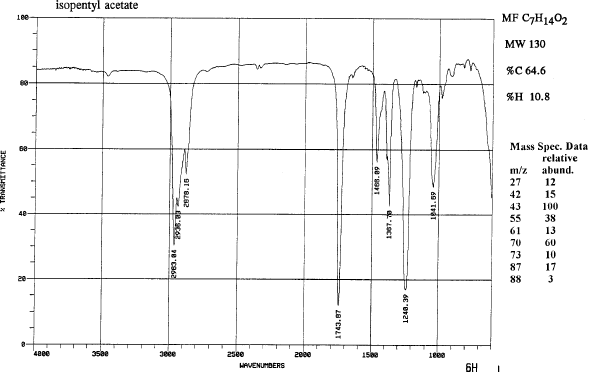
| Compound | MW | Amount | mmol | mp | bp | Density | ηD | msds |
|---|---|---|---|---|---|---|---|---|
| Isopentyl alcohol | 88.15 | 20.0 mL | 184 | 130 | 0.809 | 1.4060 | >msds | |
| Glacial acetic acid | 60.05 | 12.0 mL | 210 | 16.2 | 118 | 1.049 | 1.3720 | >msds |
| Sulfuric acid, 98% | 98.08 | 1 mL | 3 | 290 | 1.840 | > | msds | |
| Isopentyl acetate | 130.19 | 142 | 0.876 | 1.4000 | >msds | |||
| Calcium chloride (anhydrous pellets) | 130.19 | 142 | 0.876 | 1.4000 | >msds | |||
| Sodium sulfate (anhydrous) | 130.19 | 142 | 0.876 | 1.4000 | >msds | |||
| Compound | g/mol | grams or mL | 10-3 mol | oC | oC | g/mL | ηD | msds |
1Toxic/Irritant
2Corrosive; (Excess solution should be disposed of properly after the lab.)
3For 15 groups, use: