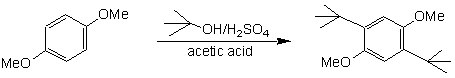
Substitution reactions are really at the heart of most organic reactions. While addition reactions occur for compounds containing either double or triple bonds, most reactions actually occur when one chemical (or atom) replaces something already on the molecule. For aromatic compounds, where the aromaticity of the compound is maintained, electrophilic substitutions take place. The electron cloud on the aromatic rings functions as the nucleophile as usually something positively charged functions as the electrophile. The mechanism of Electrophile Aromatic Substitution reactions (EAS) is not different for the many different electrophiles that could reaction. The only real difference is how to produce the appropriate electrophile for reaction. In the experiment we conduct today, we use a Friedel-Crafts Alkylation. How the alkyl cation is produced is not important. The important part is that a carbocation is used for this reaction. A t-butyl cation will be used and since no carbocation rearrangement can occur, any method can be used to produce this cation. Just remember that while AlCl3 is often used for Friedel-Craft reactions, this class of EAS simply requires a carbocation, regardless how it is generated.
We performed an Electrophilic Aromatic Substitution (EAS) reaction previously to add a nitro group to methyl benzoate in order to produce methyl m-nitrobenzoate. In the current experiment, we will use a different electrophile (the t-butyl cation) to alkylate 1,4-dimethoxybenzene. This is a Friedel-Crafts Alkylation reaction.
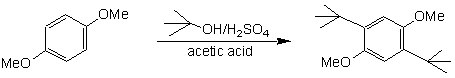
The primary difference between Friedel-Crafts EAS reactions and other EAS reactions (e.g., nitration, halogenation, etc.) is that in a Friedel-Crafts reaction (alkylation or acylation), the electrophile is a carbon atom (e.g., in this experiment it is a t-butyl carbocation). In many cases, the Friedel-Crafts electrophile is generated using a chlorine-containing molecule (e.g., an acyl [acid] chloride or an alkyl chloride) and an appropriate Lewis acid (e.g., AlCl3) catalyst (examples are shown below):
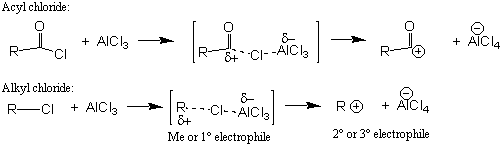
Alternatively, any stable carbocation (e.g., a 2o or 3o carbocation) can be used as an electrophile for this reaction. As we have done before, we can generate these carbocations using reactions where acid is present, as we have seen before:
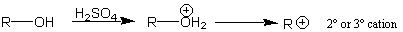
As long as the carbocation is stable, and does not undergo rearrangements, this is actually a very good way to generate an electrophile for a Friedel-Crafts alkylation reaction.
The t-butyl cation, which is used as the electrophile in this reaction, is a 3° carbocation which cannot rearrange, so it works quite well. However, as this reaction illustrates, one of the problems with Friedel-Crafts alkylations is that it is often difficult to avoid producing significant amounts of dialkylation substitution products because the alkyl group being attached to the aromatic ring is electron-donating. Therefore, when an electron releasing group is added, it activates the ring for additional alkylation reactions. The reaction we will be performing actually takes advantage of that fact, as the dialkylation product shown above is not only the desired product, but it is the only product. What factors are responsible for producing this as the only dialkylation product?
Safety: Concentrated sulfuric acid is a strong oxidizer, and both it and acetic acid are highly corrosivewear gloves while handling them, and avoid breathing their vapors. 2,5-di-t-butyl-1,4-dimethoxybenzene and 1,4-dimethoxybenzene are irritantswear gloves while handling them. Methanol is a flammable liquid, and is toxic no flames will be allowed in lab, wear gloves while handling it, and avoid breathing its vapors.
This is a small scale experiment, so it is crucial that you are careful, or you will end up with no product.
Use a dry and clean 125-mL Erhlenmeyer flask for this reaction.
Combine each of the following reagents in the reaction vessel:
Mix the above reagents together until all the solid has dissolved. When only liquid is present, you should place the flask very briefly into an ice bath to cool its contents, but do not let it sit in the ice bath too long as solids may reform. The purpose of this cooling is to prevent a large increase in temperature when the concentrated sulfuric acid is added.
Obtain about 4.0 mL of concentrated sulfuric acid and place it in an ice bath. Add the cold sulfuric acid drop wise (about 1 drop per second or slower) to the cooled reaction mixture from above. After the addition of a few drops of sulfuric acid, swirling the mixture to thoroughly mix its contents. You should keep the reaction mixture in the ice bath and swirl it occasionally during the addition of all of the sulfuric acid.
Remove the flask containing the reaction mixture from the ice bath and set in on the bench top. Allow the reaction mixture to gradually warm up to room temperature. Take your time, do not rush this step. Continue to swirl the mixture occasionally during the warming up process.
Add about 10 g of ice to the reaction mixture (which would be approximately the volume of the reaction mixture; too much ice is not a problem as it just melts). Once the ice has completely melted, add about 50 mL of DI water and mix the contents of the flask in order to precipitate the reaction product (this organic chemical is not soluble in water, so too much ice and/or water is not a problem). Isolate the solid product by vacuum filtration using a Büchner funnel. Wash the solid chemical collected on the filter with water in order to remove any water-soluble chemicals (e.g., acid and salt).
You should dry your sample in the dessicator (drying oven) until the next lab period. After your sample is completely dry (at the beginning of the next lab period), remove a small amount of the dried crude product for melt point analysis (remove a sample of the crude material at the beginning of the second day).
Day 2:
Re-crystallize the dried product using methanol as your re-crystallization solvent. Add enough methanol to dissolve the product. You do not want to add too much methanol, but if you do, you can always evaporate some of it away.
In order to facilitate your re-crystallization, do the following:
Your sample should be a free flowing slurry, meaning that there is enough liquid to keep the solid moving around. If you do not produce a lot of crystals, you might try evaporating some of the solvent by heating the mixture to the boiling point of methanol (about 68oC). Remove the beaker and let it sit again at room temperature until crystals form.
In order to collect your solid material, you will do a vacuum filtration. When you add your filter paper to the Buchner Funnel, do not wet with water, you must keep it dry. You can use a little bit of methanol to add to the filter prior to adding your solid, but do not use water. Wash your beaker with a little methanol, to attempt to add as much of your solid as possible to the funnel. Let the vacuum continue to draw air over your sample, letting it dry by evaporating all of the methanol (about 15 min). After this 15-min period of drying your sample, you can use the crystalline material directly in a melt temp. Weigh your solid and determine a yield.
To analyze your product, do each of the following:
Show the product to the instructor when you turn in your lab notebook for grading on the day you complete the experiment.
| Compound | MW | Amount | mmol | mp | bp | Density | ηD | msds |
|---|---|---|---|---|---|---|---|---|
| 1,4-dimethoxybenzene | 138.1668 | 1.00 g | 7.3 | 55-60 | 212.6 | 0.79 | msds | |
| t-butyl alcohol | 74.1224 | 2.0 mL (use pipettor) | 21.2 | 25.5 | 82.2 | 0.786 | 1.3878 | msds |
| Glacial acetic acid | 60.0524 | 3.0 mL | 52.4 | 16.6 | 117.9 | 1.0492 | 1.3719 | msds |
| Concentrated H2SO4 | 98.0734 | 4.0 mL | 75.0 | 3 | 280 | 1.84 | --- | msds |
| Methanol | 32.042 | --- | --- | -98 | 64.6 | 0.791 | 1.3286 | msds |
| 2,5-di-t-butyl-1,4-dimethoxybenzene | 250.3786 | -- | -- | 104-105 | -- | -- | -- | |
| Compound | g/mol | grams or mL | 10-3 mol | oC | oC | g/mL | ηD | msds |
Pre-lab preparation for Experiment II (this must be done during the second day of this lab).
Please note that before you leave the lab at the end of this experiment (after the second day of this lab), you must place the clean glassware that will be needed for Experiment II into the drying oven (which will be heated to remove water). The glassware must be completely dry before starting the Grignard Reaction in Experiment II. (Be sure to remove all plastic parts from your glassware, including graduated cylinder bases before heating.)
Include all of the following to be put into the oven to heat and remove water:
Go To Experiment: ChemDraw 1
2
3 4
5 6
7 8
9 10
Return to Chem211 Experiment Protocols Index
Copyright Donald L. Robertson (Date last modified: 11/14/2012)